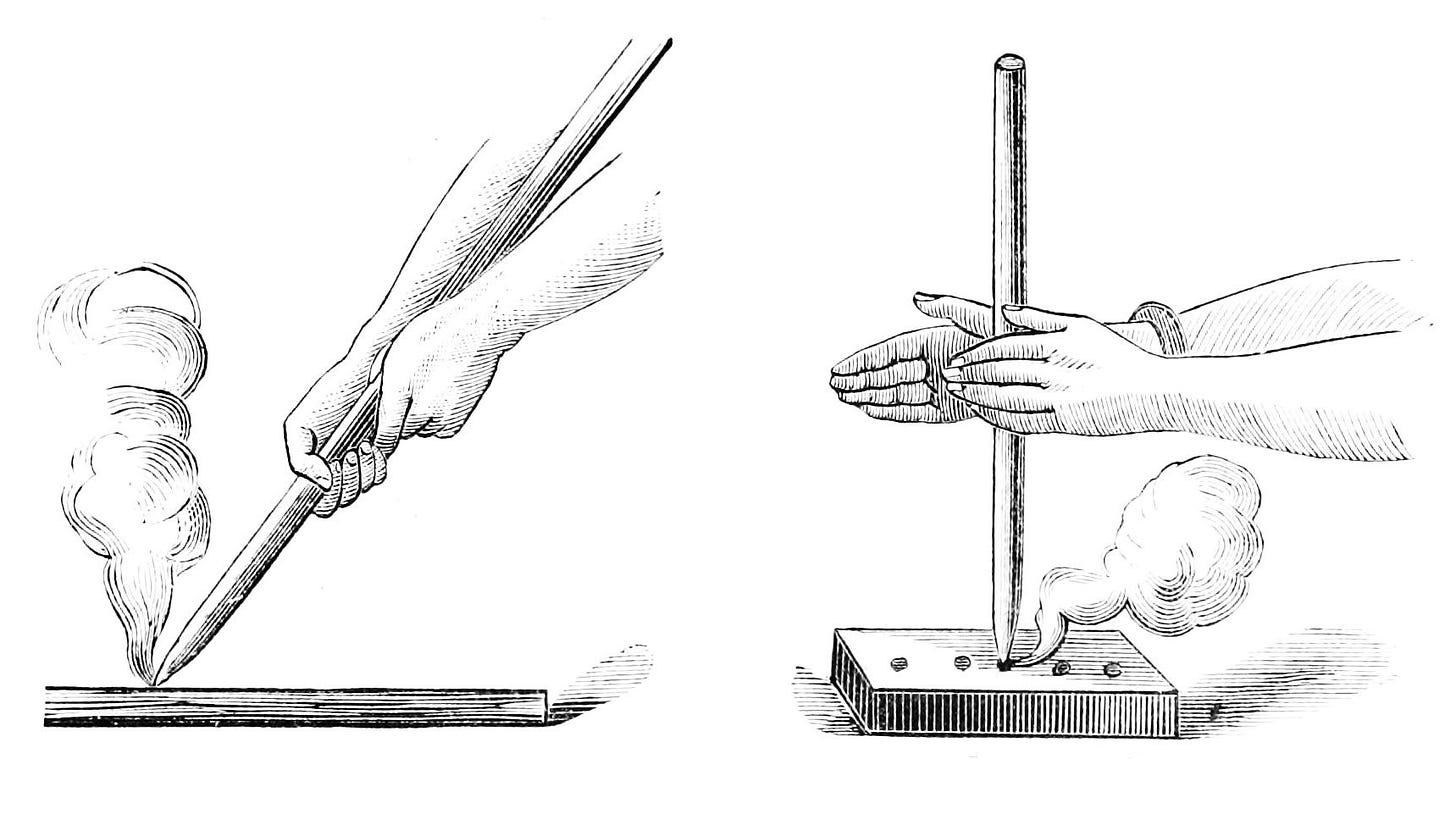
The discovery of fire and the ability to control it was a critical event in the evolution of humans. It enabled humans to eat cooked food, reduce diseases by killing organisms in food, keep predators away, make advanced hunting weapons as well as provide warmth and lighting.
Early methods
Fire occurs naturally due to lightning, forest fires, volcanic activity, and meteorites. The early ways to make a fire would have been to use the hot ashes or burning wood from a forest or grass fire, and then to keep the fire going for as long as possible by adding more combustible material.
Gradually new methods of creating fire were developed that included the use of friction (rubbing two materials against each other), the use of percussion (striking one material with another), or the use solar (focusing sun energy on a small surface).
The modern chemical method led to the development of match sticks, which enabled ‘making’ fire cheap, easy, and safe. And that’s the topic of this article!
The word match comes from the French mèche, a wick, as in a lamp. Both words
probably are derived from the Greek μυξαor the Latin myxa, mixa, or myxus, the wick of a lamp.
Answer will be shared in next newsletter.
Light-bringing Slave
The first primitive version of the “idea” of a matchstick was developed in the 6th century AD by women living in Northern Qi (China). These women were unable to leave the confines of their city to search for tinder, because the city was under siege.
So they dipped small pieces of pine into sulfur. Once the sulfur dried up, the stick would burst into flame with just a small spark (which was created by the friction method of rubbing sticks). These sulfur-dipped sticks were called "light-bringing slaves", and their later commercial name was changed to 'fire inch stick'.

One question: From where did the sulfur come?
Sulfur was often found in elemental form and also as sulfide and sulfate minerals. A natural form of sulfur known as shiliuhuang was known in China since the 6th century BC. In ancient times, sulfur was also called “Brimstone”, which means burning stone. The importance of sulfur discovery and usage will be found again during the discovery of gunpowder, which also happened in China.

Spills and Spill Vase
Around the 10th century AD, humanity was facing 2 problems in this field.
How to create fire quickly in a safe and inexpensive way.
How to transfer fire from one place to another in a safe and inexpensive way.
While the same old methods were being used as solutions to problem 1, new ways were being used for the second problem.
In the 15th century AD, the usage of the Spill Vase was common. A spill vase, in its simple form, was a cylindrical vase containing splints, spills, and tapers for transferring fire, from an already-lit fireplace to the required area. Spills were thin objects made of tightly rolled paper tapers or very thin wood sticks (splints). They could burn for a considerable time and allowed the transfer of fire to happen safely.



First self-igniting match
Many chemicals and chemical combinations were discovered that could result in fire. The flammable nature of Phosphorus was discovered by Hennig Brand in 1669, for example. But none of the methods could produce fire in a simple and inexpensive way.
In 1805, Jean Chancel invented the first modern self-igniting match, in Paris. Chancel’s match had a head that consisted of a mixture of potassium chlorate, sulfur, gum arabic, and sugar. It was ignited by dipping the head in sulfuric acid stored in a small asbestos bottle. Although a breakthrough event in this journey, Chancel’s match was dangerous and expensive. This prevented it from becoming a commonplace tool.
Prometheans and Vesuvians
Samuel Jones of London in 1828, patented another self-igniting match which was named “The Promethean match”. This had a small glass capsule containing sulfuric acid and coated on the exterior with potassium chlorate. This entire thing was wrapped up in rolls of paper.
How to achieve fire? Simple. Just take a pair of pliers, crush the capsule and mix all the ingredients. Easy peasy(?!)
In 1849, Heurtner of London invented Vesuvians, sometimes called flamers or wind matches. They were provided with a large pear-shaped head consisting generally of potassium nitrate, powdered charcoal, and wood dust all held together by glue. The head was tipped with regular phosphorus compositions and the stems were made of many materials, including wood, glass, and porcelain.
Both Vesuvians and Prometheans required a bulb of sulfuric acid at the tip which had to be broken to start the reaction. Due to their inherent danger, not-so-easy use, and high cost, such matches failed to get into mass production.
Friction Matches
John Walker, a chemist from England was working on finding the right chemical combination that would easily self-ignite. One day, in 1826, a match that was dipped in a chemical solution fell on the floor of his workplace and caught fire by accidental friction. This was a key moment of discovery and the birth of friction matches.
Walker’s match consisted of wooden splints coated with sulfur and tipped with a mixture of sulfide of antimony, chlorate of potash, and gum. Camphor was added to improve the odor. It was packed in a box of 50 matches and a piece of sandpaper, (double-folded) was provided with each box.

How to achieve fire? Draw the match through the folded sandpaper to ignite it.
What a progress!!
Between 1827 and 1829, Walker sold 168 boxes of his friction match. The price of a box of 50 matches was one shilling. (around $4.2 in 2022)
While the problem of ease of ignition was almost solved, the problem of safety was still there. Walker’s matches were dangerous and flaming balls sometimes fell to the floor burning carpets and dresses, leading to their ban in France and Germany.
Lucifer’s Matches
In 1829, Sir Isaac Holden improved upon Walker’s match and Samuel Jones patented a modified version of Holden’s match. It was then sold under the name of “Lucifer match”. The lucifer match heads were composed of equal quantities of potash of chlorate, sulfide of antimony and glue.
Despite all improvements (in the speed of ignition, handling safety, etc.), the matches were still dangerous overall. Lucifer matches would often scatter burning fragments and the ignited match head would not always ignite the entire matchstick.
The Phosphorus Saga
The entire history of matches took a significant turn when Charles Sauria, a French chemist substituted the sulfide of antimony in Lucifer matches, with White phosphorus in 1830.
Phosphorus is itself not the fuel but acts as the initiator for the flame. It readily oxidizes in air, but the glue in the lucifer match-head proved to be sufficient protection against premature oxidation. The new match was comprised of white phosphorus (20%), sulfur (15%), potash of chlorate (30%), alum (10%), and glue (25%).
Initially, phosphorus was considered as a fuel element for the match, but when its role as the initiator was realized the phosphorus content of the match was reduced, and only one milligram of phosphorus per match is necessary.
Phosphorus matches were sold under the name of loco foco in United States.
For the next 60 years, the chemical composition of the matches remained nearly the same. Some small improvements were made for different purposes like —
In 1836, János Irinyi a Hungarian Chemist invented a noiseless match, by replacing the chlorate of potash with lead dioxide.
In 1843 William Ashgard replaced the sulfur with beeswax, making the fumes less pungent. It was further replaced by paraffin in 1862 by Charles W. Smith and was sold as “Parlor Matches”.
In 1870 the end of the splint was fireproofed by impregnation with fire-retardant chemicals to prevent the accidental burning of the user's fingers. These were sold as “Drunkard’s matches”.
And then there was… Darkness!
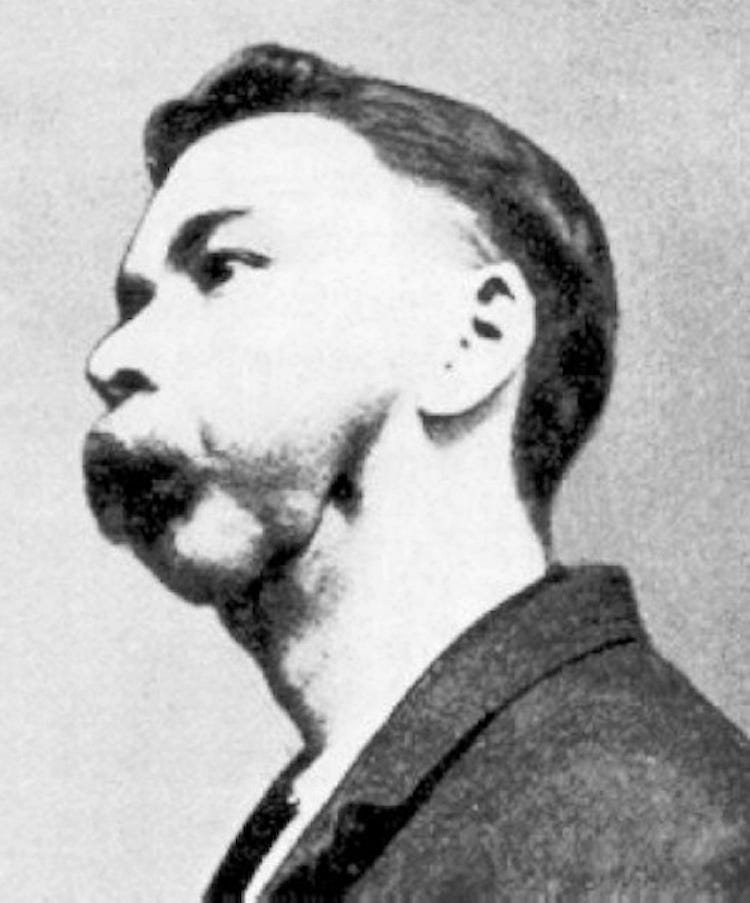
All the goodness of white phosphorus soon came to a big halt when it was discovered as the main cause of a very painful and horrific disease: Phossy Jaw. It was caused by white phosphorus vapor, which destroyed the bones of the jaw and caused serious complications.
The initial symptoms of Phossy Jaw included toothaches and swelling of gums. Within a few weeks it deteriorated to pus formation, recurrent abscesses, formation of sequestrum (a dead bone that has separated from living bone), and disfigurement of the lower part of the face. The disease was extremely painful and depressing to the patient which was made worse by the social stigma and loss of employment. Suicides, depression, and deaths from this disease happened in many places.
The first case of the phossy jaw was diagnosed by physician Lorinser of Vienna in 1839. The patient was a female Viennese matchstick maker who had been exposed to phosphorus vapors over a five-year period.
Matchgirls' strike
Bryant & May, a prominent British match-making company was aware of the phossy jaw. If a worker complained of having a toothache, they were told to have the teeth removed immediately or get fired.
Social activist Annie Besant published an article in her halfpenny weekly paper The Link on 23 June 1888, highlighting the plight of the women workers.

Bryant & May management tried to get their workforce to sign a paper contradicting the article, but the workers refused. A worker got dismissed and this triggered the strike with approximately 1,400 women and girls refusing to work and by the 6th of July the whole factory had stopped working. The strike and negative publicity led to changes being made to limit the health effects of the inhalation of white phosphorus.

It is banned!
White phosphorus continued to be used, and its serious effects led many countries to ban its use. Finland was the first to ban its use in 1872, followed by Denmark in 1874, France in 1897, Switzerland in 1898, and the Netherlands in 1901.
An agreement, the Berne Convention, was reached in Bern, Switzerland, in 1906, which banned the use of white phosphorus in matches. The United Kingdom banned it in 1910, Canada in 1914, India and Japan in 1919, and China in 1925.
The United States did not pass a law, but instead placed a very high "punitive tax" in 1913 on white phosphorus–based matches.
Going from White to Red
Anton Schrötter von Kristelli discovered the red allotropic form of phosphorus in 1850. (Allotropy is the property of a chemical element to exist in two or more different forms, in the same physical state).
This allotropic form did not produce fumes in contact with air and was made by heating white phosphorus at 250 °C in an inert atmosphere.
While it could be used as a good substitute in matches, the overall manufacturing was expensive. To compete with the price of white phosphorus matches, many companies including Bryant and May, started using cheap child labor extensively.
Then in 1898 two French chemists, Henri Savene and Emile David Cahen, discovered phosphorus sesquisulfide which was much less toxic and less explosive in nature. British company Albright and Wilson was the first company to produce phosphorus sesquisulfide matches commercially.
In the United States, the Diamond Match Company got a patent for a non-poisonous match that used sesquisulfide of phosphorus. U.S. President William Howard Taft requested that Diamond Match give up its patent which the company agreed.
Safety Matches
The safety of true "safety matches" is derived from two aspects.
The separation of the reactive ingredients between a match head and a special striking surface. (Remember the previously discussed matches were strike-anywhere friction matches).
Replacing white phosphorus with red phosphorus.
After Schrötter had discovered the red allotrope, Arthur Albright developed the industrial process for manufacturing it at a large-scale in the 1850s. Gustaf Erik Pasch, a Swedish professor of chemistry, developed the idea of a separate striking surface and patented the use of red phosphorus in it.
Johan Edvard Lundström and his younger brother Carl Frans Lundström started large-scale manufacturing of safety matches with red phosphorus-based striking surfaces. By 1858, their company produced around 12 million matchboxes.
These types of matches were large and had to be carried in a wooden box. There was a person who didn’t like this thing. He was Joshua Pusey, an American inventor and an attorney. He invented paper matches that would be lighter and smaller. His final patented design had matches secured to a thin paper wrapping with an attached striking surface.

Pusey later sold the patent for the matchbook (which he called flexibles) to the Diamond Match Company for $4000 in 1896. (~$140,000 in 2022).
Pusey had positioned the striking surface on the inside of the paper fold which allowed all 50 matches to be lit at once. The design for a modern matchbook with the striking surface on the outside of the box was patented by Charles M. Bowman. He also sold his patent to the Diamond Match Company in 1902.
How do safety matches work?
The striking surface on modern safety matchboxes is typically composed of 50% red phosphorus. The remaining is a mixture of abrasives, neutralizers, carbon black, and binder.
The match head is typically composed of 50% potassium chlorate, with a little sulfur and starch, a neutralizer, filler, stabilizer, and glue
When a safety match is struck, the red phosphorus from the striking surface mix with potassium chlorate in the match head in a small amount. This mixture ignites due to friction.
Good to Know
The strike-anywhere friction matches made with white phosphorus as well as those made from phosphorus sesquisulfide are still widely used for uses in outdoor activities/emergency kits. However, it is banned on all kinds of aircraft under the "dangerous goods" classification.
Modern times have brought Storm matches, also known as lifeboat matches or flare matches. These can burn strongly even in strong winds, and can even spontaneously re-ignite after being briefly immersed under water. We have also got matches that can burn underwater!

Special Storm Matches. Credits: By Fornax The hobby of collecting match-related items, such as matchcovers and matchbox labels, is known as phillumeny.
So, that was the journey taken by humanity in development of a matchstick.
Next time you ignite one, do give a thought on how much trouble and cumulative hard work humans have put into this seemingly simple beauty!
Feel free to ask any questions or share your thoughts on this article or any feedback you have. This publication is for made for you!